Global pharma companies today face a critical challenge – accelerating innovation and strengthening pipelines while navigating complex regulatory environments and cost pressures. Key priorities include:
The key factors that have made India the backbone of global life sciences GCC operations include:
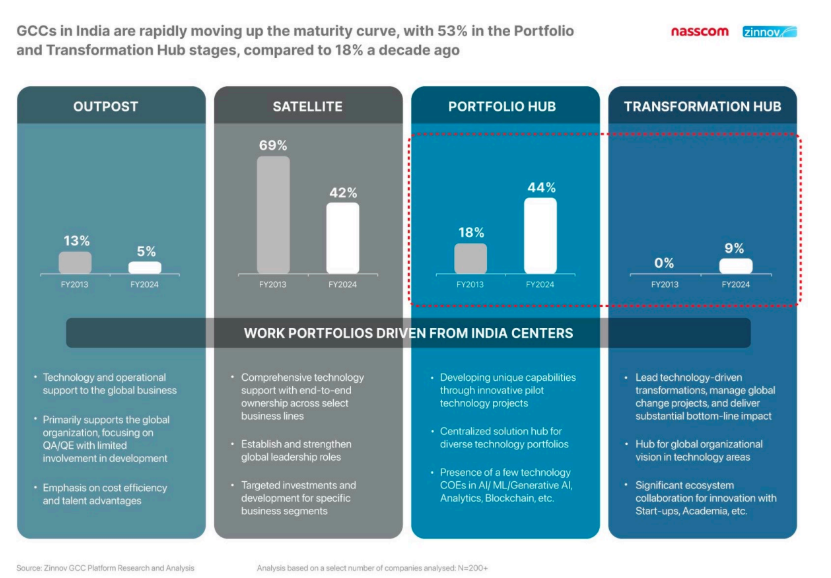
Today’s GCCs aren’t back-office operations. They’re innovation hubs contributing directly to drug discovery, digital therapeutics, and patient-centric breakthroughs.
Outsourcing providers simplify workforce capacity, but add 40-60% gross margins to every element. You lose visibility into actual costs, control over quality and processes, and cultural alignment between your strategic priorities and operational execution.
Direct cost control, full operational authority, and sustainable capability building, while optimizing spend by up to 25%. Modern GCCs are strategic innovation engines, not cost centers, contributing directly to pipeline acceleration and competitive differentiation.
Building a GCC isn’t just about hiring people and leasing office space. It’s about creating a sustainable capability that meets global regulatory standards, delivers predictable outcomes, and scales without quality erosion.
| What You Get | Life Sciences GCC | Outsourcing Provider |
|---|---|---|
| Cost Structure | ~25% cost optimization | 40–60% gross margins embedded |
| Cost Visibility | Full transparency across direct & indirect costs | Hidden markups, bundled fees |
| Operational Control | Complete control over SOPs & quality | Governed by vendor processes |
| Compliance Oversight | Direct audit documentation control | Vendor-dependent compliance |
| Talent Access | Hire from open market or trained pool | Restricted to provider bench |
| Long-Term Stability | Build internal capability | Locked into vendor contracts |
| Scalability | Predictable, business-aligned scaling | Vendor capacity & pricing dependent |
| Cultural Alignment | Mirrors HQ ethos & strategy | Operational satellite model |
| IP Ownership | Fully protected internally | Shared exposure risk |

Consulting on functional scope, role structures, SOP alignment, operating models, governance frameworks, program management and KPI design for Pharmacovigilance, Clinical Operations, Quality Assurance, Regulatory Affairs, and Medical Writing GCCs.

Structured sourcing strategies and readiness programs to ensure consistent skill quality, compliance awareness, and delivery readiness across teams.

Site Selection, Workplace Design & Fit-Out Planning and Facilities & Operations Planning.

Technology Assessment & design coupled with Process Optimization, Automation and CoE establishment

Training and evaluation frameworks built to meet global regulatory expectations, audit standards, upskilling, and governance mechanisms and client delivery requirements.

People
Functional Talent strategy, hiring, competency frameworks
Process
Operating model design, SOP architecture, productivity benchmarking, governance frameworks

Technology
Digital enablement, automation, AI-led productivity, compliant tech stack selection

Infrastructure
Location strategy, facility design, compliance-ready infrastructure, scalability planning
Project management expertise for seamless execution across all pillars

ExpertiseInPharma is a global organization dedicated to empowering pharmaceutical sponsors and contract research organizations (CROs) navigate complex regulatory landscapes, accelerate innovation and boost productivity.
Our solutions, including Global Capability Center (GCC) Consulting, SME on Demand, Resource on Demand and Outsourced L&D, are curated and delivered by industry practitioners who understand pharma delivery environments, regulatory expectations, and execution pressures, thus helping clients convert capability into measurable outcomes.